Batch Release without Determination of Identity and Strength and other GMP violations - A Look at FDA's Warning Letters over the Last Months
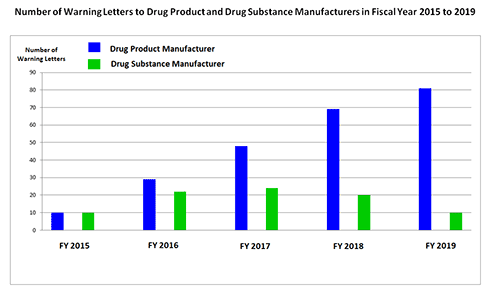
When analysing the Warning Letters sent to manufacturers of finished medicinal products worldwide in the last fiscal year (from October 2018 to September 2019), it is first of all the sheer amount that surprises: with a total of 81 Warning Letters, this is the highest number over a five-year period (the Warning Letters addressed to Compounders or Compounding Pharmacies are not included in this analysis). In contrast, the number of Warning Letters to API manufacturers has fallen to the level of 5 years before after a peak value in fiscal year 2017.
Plus d'information ici